Molnupiravir Merck Kaufen, Usa Planen Milliardenschwere Bestellung Von Coronamedikament
Merck on Friday announced that its new pill to treat Covid-19 reduced the risk of hospitalization and. In anticipation of the results from MOVe-OUT Merck has been producing molnupiravir at risk.

Merck Announces Supply Agreement With U S Government For Molnupiravir An Investigational Oral Antiviral Candidate For Treatment Of Mild To Moderate Covid 19 Merck Com
Molnupiravir has been shown to be active in several models of SARS-CoV-2 including for prophylaxis.

Molnupiravir merck kaufen. Wie es das Coronavirus stoppt war bisher unklar. Die Einnahme von Molnupiravir habe das Risiko im Krankenhaus behandelt werden zu müssen um die. About Mercks Efforts to Enable Access to Molnupiravir if it is Granted EUA or Approval.
If it gets authorization molnupiravir which is designed to introduce errors into the genetic code of the virus would be the first oral antiviral. Eigentlich sollte es ein Mittel gegen Grippe werden. Molnupiravir EIDD-2801MK-4482 is an investigational orally bioavailable form of a potent ribonucleoside analog that inhibits the replication of multiple RNA viruses including SARS-CoV-2 the causative agent of COVID-19.
All you need to know about Mercks COVID-19 oral pill. Die Firmen MSD und Ridgeback Biotherapeutics haben nach einer Zwischenanalyse angekündigt eine Phase-III-Studie bei nicht hospitalisierten Covid-19-Patienten fortzusetzen. Mercks experimental pill molnupiravir is the first oral antiviral medicine intended to treat COVID-19.
Merck and Ridgeback Biotherapeutics today announced that molnupiravir MK-4482 EIDD-2801 an investigational oral antiviral medicine significantly reduced the risk of hospitalization or death. By Sissi Cao 100121 1205pm If approved molnupiravir will be the first antiviral. Laboratory studies show that Merck Cos experimental oral COVID-19 antiviral drug molnupiravir is likely to be effective against known variants of the coronavirus including the dominant.
Earlier this year Merck entered into a procurement agreement with the US. Merck Cos molnupiravir the Covid-19 pill claims to help nations which are struggling to vaccinate their citizens. If molnupiravir receives regulatory support physicians themselves will likely determine treatment eligibility based on each patients risk-benefit profile University of North Carolina infectious disease expert Dr Myron Cohen.
Es handelt sich um einen sogenannten Polymerasen-Hemmer. Mercks Molnupiravir EIDD-281 Effective Against SARS-COV-2 Molnupiravir is a nucleoside analog. Beth Mole - Oct 1 2021 504 pm UTC.
High-risk individuals likely first users. It has shown to be able to cause mutations. According to an analysis based on a late-stage trial conducted by Merck and.
RTTNews - Drug major Merck Co Inc. Merck called MSD outside the US and Canada has initiated a rolling submission to Health Canada for molnupiravir an investigational oral antiviral therapy for the treatment of Covid-19. Molnupiravir befindet sich derzeit in Phase 3 der klinischen Tests.
Meet molnupiravir Mercks Thor-inspired pill that hammers COVID In Phase III trial the drug smashed hospitalization and death rate by about half. Merck expects to produce 10 million courses of treatment by the end of 2021 with more doses expected to be produced in 2022. Being developed in collaboration with Ridgeback Biotherapeutics molnupiravir is an investigational orally twice administered antiviral candidate that is currently in trials as a potential treatment for.
Der Pharmakonzern Merck meldet positive Ergebnisse einer Studie zu einem neuen Corona-Medikament. Government under which Merck will supply approximately 17 million. Es wirkt besser als Remdesivir die USA kaufen von dem Medikament Dosen für eine Milliarde Dollar.
Göttinger Forscher haben Molnupiravir untersucht. Jetzt haben Göttinger. Merck Co Inc and partner Ridgeback Biotherapeutics said on Wednesday they had begun enrolling patients in a late-stage trial of their experimental drug molnupiravir for prevention of COVID-19.
Earlier this year Merck and Ridgeback reported preliminary data from a Phase IIa Covid-19 trial of molnupiravir in the US. MRK and Ridgeback Biotherapeutics Friday announced positive interim analysis of Phase 3 MOVe-OUT. USA wollen experimentelles Corona-Medikament Molnupiravir kaufen.
An experimental antiviral therapy molnupiravir is an oral form of a potent ribonucleoside analogue that hinders the replication of various ribonucleic acid RNA viruses such as SARS-CoV-2. Molnupiravir tricks the coronavirus into using the drug to. Merck MSD and Ridgeback Biotherapeutics have commenced a Phase III MOVe-AHEAD clinical trial of their oral drug molnupiravir to prevent Covid-19.
Merck and its partner Ridgeback Biotherapeutics are currently assessing the drug in a global Phase III trial to treat non-hospitalised Covid-19 patients. Merck is developing molnupiravir in collaboration with Miami Florida-based Ridgeback as per a deal announced in May 2020. Tatsächlich zeigt sich Molnupiravir wirksam gegen Sars-CoV-2.
Merck on Friday announced its anti-viral drug has shown compelling results in clinical trials halving the risk of hospitalisation or death for patients with mild or moderate cases of Covid. Merck and its partner Ridgeback Biotherapeutics said early results showed patients who received the drug called molnupiravir within five days of COVID-19 symptoms had about half the rate of. Merck MRK and Ridgeback Biotherapeutics announce results from their oral antiviral medicine molnupiravir MK-4482 EIDD-2801 from Phase 3 MOVe-OUT trial in patients.
Der antivirale Wirkstoff Molnupiravir ist derzeit als mögliches Covid-19-Medikament im Gespräch. Molnupiravir co-developed with Ridgeback Biotherapeutics is. Meanwhile Mercks molnupiravir could halve the chances of dying or being hospitalized for those most at risk of contracting severe COVID-19 according to data that experts hailed as a potential breakthrough in how the virus is treated.

Molnupiravir The Drug That May Completely Stop The Spread Of Coronavirus In 24 Hours

U S Signs 1 2 Billion Deal For 1 7 Million Courses Of Merck S Experimental Covid Drug

Biden Administration Plans To Purchase 1 7 Million Courses Of Merck S Antiviral Treatment Thehill
Merck Nabs 1 2 Billion Order For Oral Covid Antiviral From Us

Covid 19 Us To Buy 1 7 Million Courses Of Merck S Antiviral Pill The New Indian Express

Usa Planen Milliardenschwere Bestellung Von Coronamedikament

Merck Announces Supply Agreement With U S Government For Molnupiravir An Investigational Oral Antiviral Candidate For Treatment Of Mild To Moderate Covid 19 Merck Com

Antiviral Drug Molnupiravir Blocks Covid 19 Virus Within 24 Hours Study Health News Et Healthworld

Anti Covid Pille Fur Zu Hause Praparate Wecken Hoffnung
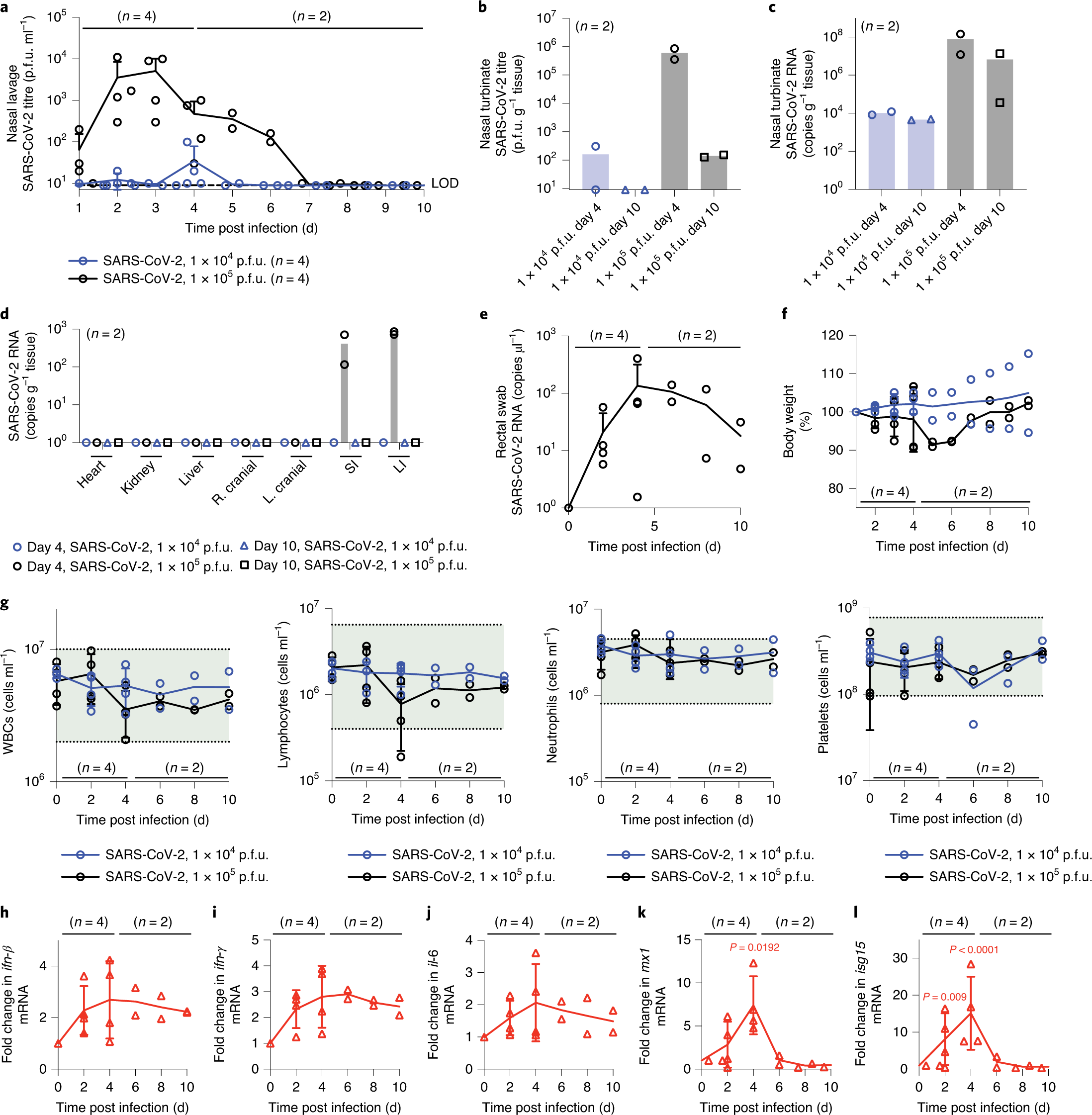
Therapeutically Administered Ribonucleoside Analogue Mk 4482 Eidd 2801 Blocks Sars Cov 2 Transmission In Ferrets Nature Microbiology

Merck Announces Supply Agreement With U S Government For Molnupiravir An Investigational Oral Antiviral Candidate For Treatment Of Mild To Moderate Covid 19 Merck Com

Merck Announces Supply Agreement With U S Government For Molnupiravir An Investigational Oral Antiviral Candidate For Treatment Of Mild To Moderate Covid 19 Merck Com

Merck Announces Supply Agreement With U S Government For Molnupiravir An Investigational Oral Antiviral Candidate For Treatment Of Mild To Moderate Covid 19 Merck Com




